The market for cellulite reduction is high and has been previously served by non-invasive devices such as the Reaction by Viora, Velashape, and Smoothshapes. The newest addition to the battle against cellulite is Cellulaze, which differs from the non-invasive procedures by using laser technology. Under local anesthesia, a small cannula is inserted under the skin and advanced SideLight 3D? technology directs controlled laser energy to treat cellulite.
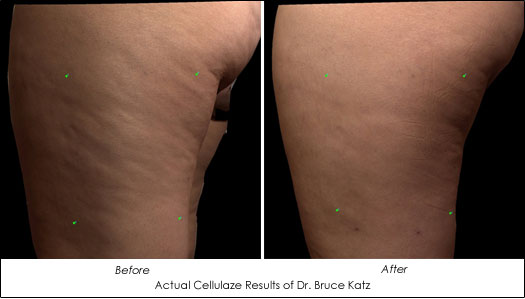
Actual Cellulaze Results of Dr. Bruce Katz
"With this revolutionary new Sidelight 3D optical fiber and Cellulaze laser, we are getting long lasting improvement in cellulite with only one treatment. This should be a major leap forward for the body contouring industry," says Dr. Bruce Katz, Clinical Professor of Dermatology at The Mount Sinai School of Medicine, Director of the Cosmetic Surgery & Laser Clinic at Mount Sinai Medial Center, and Director of the Juva Skin & Laser Center in New York.
Designed to treat lumpy pockets of fat by releasing areas of skin depression and increasing elasticity and thickness of skin, the Cellulaze is one of the first lasers to hit the market to address cellulite. An added benefit is patients only require one treatment.
Dr. Katz, a pioneer of SmartLipo and one of the leading physicians working on Cellulaze for cellulite explains, "Through our Vectra Analysis scans, we can actually measure the skin's topography and demonstrate how cellulite is reduced after patients undergo the Cellulaze procedure. This is the first time scientifically proven results for the reduction of cellulite has been recorded. We have seen excellent results with Cellulaze in its trials here in the U.S." 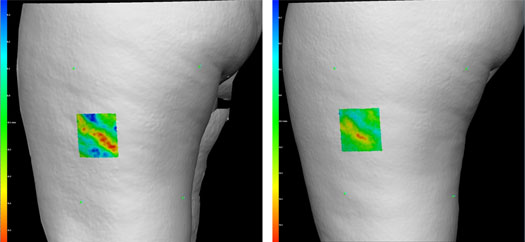
Actual Cellulaze Results of Dr. Bruce Katz
Vectra Analysis Scan
Cellulaze has received its CE Mark certification in Europe and will be available to physicians in the European Union in the second quarter of 2011. It will first be available in France, Germany, Spain, and the UK. While not yet available in the US, the FDA has approved an Investigational Device Exemption ("IDE") for Cellulaze, and nearly 100 patients have been treated as part of a clinical IDE study.
For additional information on Cellulaze, please contact Juva Skin & Laser Center in New York at (212) 688-5882.