Cynosure announced today receiving FDA clearance for it's newest cellulite treatment device the Cellulaze.
The cellulite reduction market is huge and the currently available treatments only work to hide the appearance of cellulite, not actually treat it. The newest addition to the battle against cellulite is the Cellulaze, which differs from the non-invasive procedures by using laser technology to actually treat the causes of Cellulite, not just hide it. Cynosure President and Chief Executive Officer Michael Davin stated in a press release, "Cellulaze is the only aesthetic device that directly treats the physiological structure of cellulite, providing clinically proven, results."

Model Photo
Cellulaze reduces cellulite - in just one treatment - by restoring the normal structure of the skin and underlying connective tissue. Cellulaze diminishes the lumpy pockets of fat, releases the areas of skin depression typifying cellulite and increases the elasticity and thickness of the skin. Nearly four years of clinical research puts Cellulaze in a class of its own: the only single-treatment aesthetic procedure clinically shown to reduce cellulite. Clinical data has demonstrated that Cellulaze treatments can increase the thickness of the skin by 25% and increase the elasticity of the skin by 29% at one year. No other anti-cellulite treatment has been proven to deliver these results.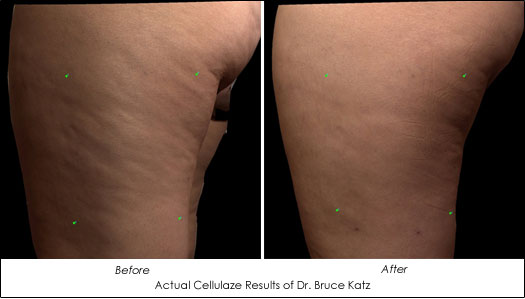
Actual Cellulaze Results of Dr. Bruce Katz
Dr. Bruce Katz, founder of the JUVA Skin & Laser Center and one of the original pioneers of SmartLipo, has been the sole dermatologist in the U.S. to conduct the clinical trials of Cellulaze that have led to its recent FDA clearance. Dr. Katz explained to American Health and Beauty, "Through our Vectra Analysis scans, we can actually measure the skin's topography and demonstrate how cellulite is reduced after patients undergo the Cellulaze procedure. This is the first time scientifically proven results for the reduction of cellulite has been recorded. We have seen excellent results with Cellulaze in its trials here in the U.S."
The 510(k) FDA Clearance is for treating "Cellulite Appearance", specifically saying the device is designed to treat cellulite appearance with the delivery of laser energy via a cannula into the sub-dermal area to dissolve fat and disrupt the septae that affect the appearance of cellulite. There are no claims as to curing cellulite in the patient or if the results are temporary or permanent.